 So the orbital numbers of " Ar" (= 3 × waveslength ), " Xe" (= 5 × waveslength ) remain the same as " Ne" and " Kr".Īn integer times de Broglie wavelength is needed The odd numbers of "3", "5", "7" orbits are asymmetrical and unstable. Radon (= Rn ) with 6 × de Broglie wavelength has the maximum " 8" orbits. Krypton (= Kr ) with 4 × de Broglie wavelength has the maximum " 6" orbits. The maxium number of orbits in Ne with 2 × de Broglie wavelength becomes " 4" (= each 2 electrons × 4 orbits = 8 valence electrons ). (Fig.3) Maximum numbers of orbits in noble gasesĪs shown on this page, de Broglie wavelength influences the number of maximum orbital number in the periodic table. So meaningless and useless.įor example, though magnesium and singly-ionized alminum have exactly the same valence electrons, their orbital orders are chaotic.ĭe Broglie "wavelength" determines the maximum orbits. They just choose fake trial functions giving their desired artificial values. Schrodinger equation has NO ablity to predict energy levels in multi-electron atoms. In one-electron hydrogen atom, energy levels are higher in larger quantum number ( 3s < 4d < 4f < 5d < 6s ).īut in other multi-electron atoms, their orders are reversed ( 4s < 3d, 6s < 4f < 5d ) ! Quantum mechanical orbital are self- contradictory in periodic table. (Fig.2) 4d and 3s, 6s and 4f orbitals are reversed in quantum mechanics !? In atoms just after noble (= inert ) gases, ionization energies suddenly drop, which indicates their electrons enter outer new different orbitals.įrom this change, we can know true numbers of valence electrons in each atom ( He = 2, Ne, Ar = 8, Kr, Xe = 18, Rn = 32 electrons ). How can we know how many valence (= outer ) electrons each atom has ? We should look at the periodicity of the first ionization energies of atoms. 
(Fig.1) ↓ Periodicity of ionization energies shows true valence electron number Top wave True numbers of atomic valence electrons Now in this electron configuration of krypton, we have to see the total number of electrons present in the highest energy level.What determines valence ( outer ) electron number ? Valence electron number 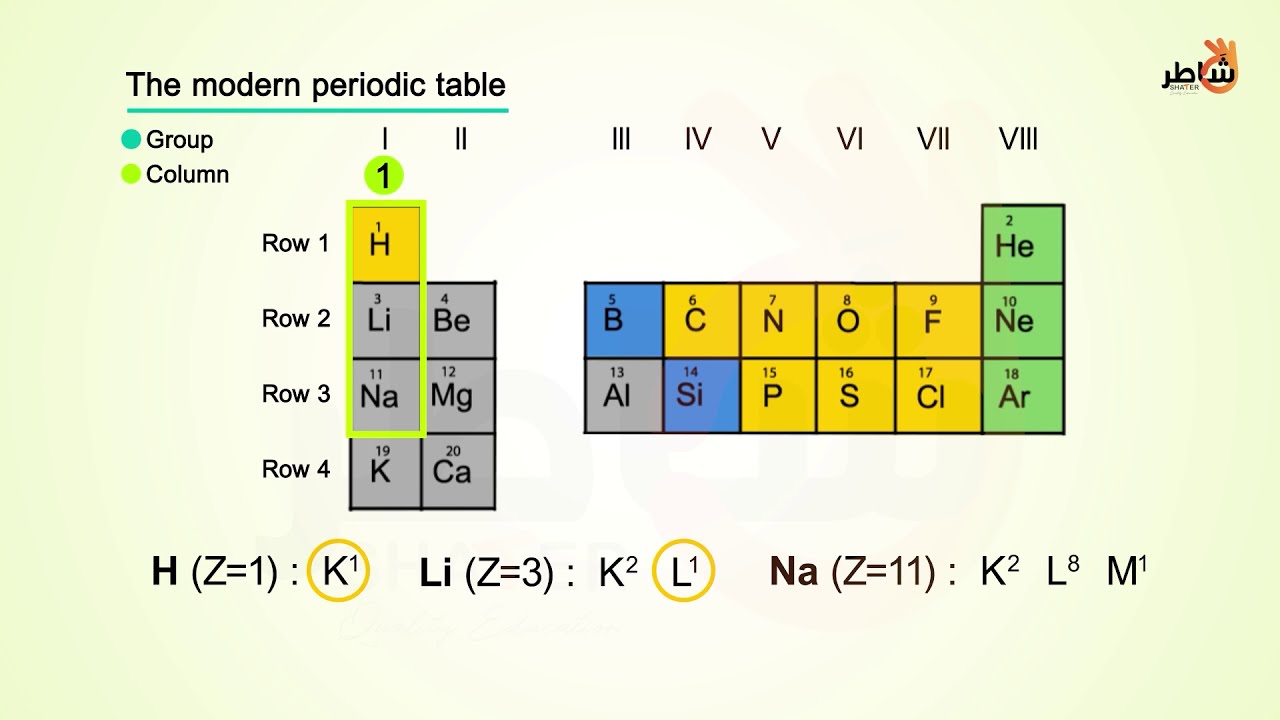
So from the Aufbau principle, we can get the electron configuration of the krypton atom as 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 2 4p 6.According to the Aufbau principle, the electrons will be filled first in 1s orbital, then in 2s orbital, then in 2p orbital, and so on…. 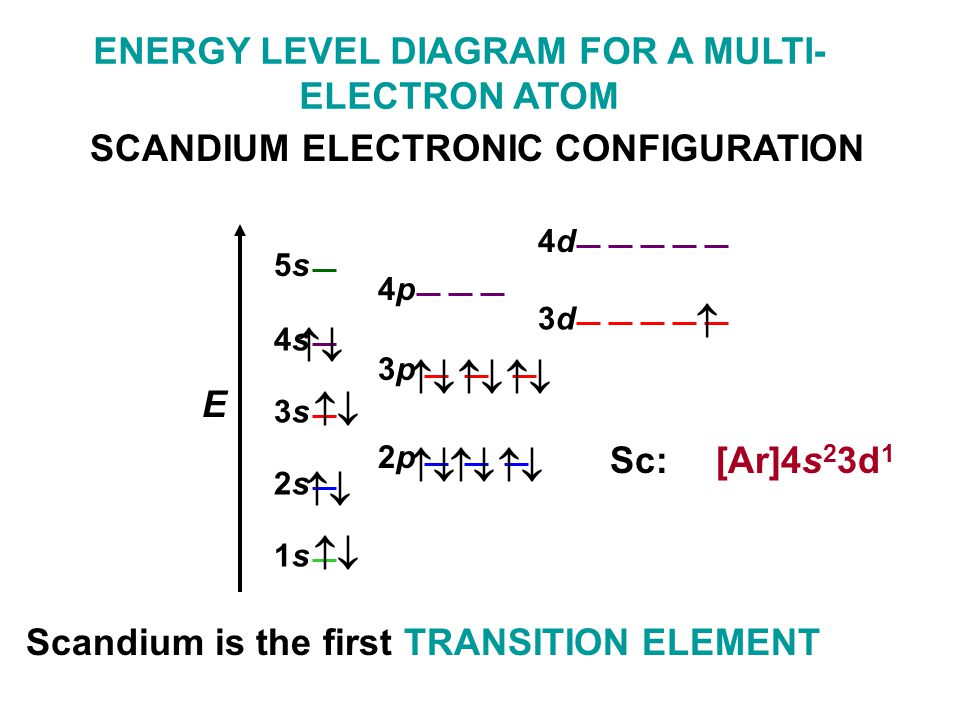
Now we have to fill these 36 electrons in the atomic orbitals according to the Aufbau principle.The krypton atom has a total of 36 electrons because its atomic number is 36 and it is a neutral atom.To write the electron configuration of krypton, we should first know the total number of electrons present in a krypton atom.Now let’s try to find the electron configuration of Krypton by using the Aufbau principle.įollow the steps mentioned below to get the electron configuration of Krypton. Orbitals Maximum capacity of electrons s 2 p 6 d 10 f 14 Now there are many methods to write the electron configurations, but here I will show you the easiest method, i.e by using Aufbau principle.Īufbau principle: The Aufbau principle simply states that the orbitals with the lower energy are filled first and then the orbitals with higher energy levels are filled.Īccording to the Aufbau principle, the orbitals are filled in the following order:ġs, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p, and so on.Īlso the maximum number of electrons that can be accommodated in s, p, d & f orbitals are mentioned in the below table. If you want to find the valence electrons of krypton from its electron configuration, then you should know its electron configuration first. Method 2: From the Electron Configuration Now let’s see another method for finding the number of valence electrons in krypton. In this way, by knowing the position of krypton element in periodic table, you can easily find its valence electrons. So, as the krypton element is present in group 18, it has 8 valence electrons. (Note: Group 18 is also called group 8A). 
More specifically, you have to see the group wise position of Krypton element in the periodic table.įrom the above image, you can see that the Krypton (Kr) is present in the group 18 of periodic table. To find out the valence electrons of Krypton, you have to see the position of krypton in the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
